All published articles of this journal are available on ScienceDirect.
Clinical and Genetic Analysis in Pediatric Patients with Multiple Sclerosis and Related Conditions: Focus on DR Genes of the Major Histocompatibility Complex
Abstract
Introduction:
There are several diseases recognized as variants of MS: post-infectious acute disseminated encephalitis, multiple sclerosis (MS), Rasmussen leukoencephalitis and Schilder's leukoencephalitis and related, but separate neuroimmune condition – Neuromyelitis Devic’s. In Kazakhstan diagnosis of such diseases was rare and immune modified treatment was only admitted after the age of 18. Clinical and immunogenetic study of MS spectrum diseases in Kazakhstan would allow to justify early targeted treatment.
Objective:
The aim of the study was to investigate genes of the main complex of human histocompatibility (MHC) associated with diseases of MS spectrum in Kazakhstani population.
Methods:
Complex clinical, neuroimaging and immunogenetic studies were performed in 34 children (24 girls, 10 boys) aged 4 to 18 years. 21 children were diagnosed with MS (11 Kazakh origin and 10 – Russian; 4 boys, 17 girls), 7 with leucoencephalitis (all Kazakh, 5 boys, 2 girls) and 6 with Devic neuromyelitis optica (all Kazakh, 1 boy, 5 girls). Genotyping of HLA DRB1, DQA1, DQB1 genes was performed for all patients.
Results:
MS group was characterized by classical relapsing-remitting MS. Predominant haplotype as a linkage complex was DRB1*15:01~DQA1*01:02~DQB1*06:02 in 20 (47.6%) of 42 DR-alleles, in 16 (76.2%) patients. MS relative risk (RR) was 13,36 for ethnic Kazakhs and RR=5,55 in Russians.
Leukoencephalitis had 7 children, with 28.6% mortality rate. The haplotype DRB1*15:01~DQA1*01:02~DQB1*06:02 as a linkage complex was detected 3 patients (4 alleles), RR=5,88.
Devic’s neuromyelitis optica (NMO) clinical course was characterized by fast and prolonged progression. There was predominance of DRB1*14 allele with RR=3,38.
Conclusion:
Summarizing, in the Kazakh population the haplotype DRB1*15:01∼DQA1*01:02∼DQB1*06:02 as a linkage complex was associated with prediction to MS and leukoencephalitis, but not to Devic’s NMO. Our study highlights the importance of awareness of MS and related disorders diagnosis which allows to implement early admission of disease-modified treatment in pediatric MS in Kazakhstan.
1. INTRODUCTION
Immune-mediated diseases of the central nervous system (CNS) in childhood are a heterogeneous group of rare conditions sharing the inflammatory involvement of the CNS [1]. They include several disorders, such as multiple sclerosis (MS), acute disseminated encephalo-myelitis, Rasmussen encephalitis and neuromyelitis optica (NMO) [2, 3]. Similar to all autoimmune conditions, children with autoimmune neurological disorders are likely to harbor some genetic predisposition in the major histocompatibility complex (MHC). The human leukocyte antigen (HLA class II) is the human version of the MHC, a gene family encoding cell-surface proteins responsible for the regulation of the immune system [2-4]. This HLA class II molecules are coded by DR-genes and expressed on the surface of macrophage (microglia in CNS), defining interaction with CD4+ T-cells during antigen presentation (autoantigen). Surface antigen presentation initiates the immune process, further leading to autoimmune diseases, for example, MS and related disorders. DR-genes (DRB1, DQA1, DQB1 and others) are inherited as a linkage complex, and every gene is coded by 2 alleles (one from parent 1 and one from parent 2).
The majority of studies showed an association of disease appearance with DRB1*15:01~DQA1*01:02~DQB1*06:02 as a linkage complex [4, 5]. In large American studies from pediatric MS centers, it was shown that HLA-DRB*15 and DQA*02 were strongly associated with the pediatric onset of MS [5, 6] and female gender [6]. The haplotype DRB1*15:01 did not influence the clinical severity but predisposed to the chronic, relapsing-remitting course of the disorder [7, 8]. Another study showed that HLA-DRB1*15:01 was equally associated with teenagers and people older than 50 years affected by MS, however, HLA-DRB1*0801 was associated more with elderly age [9]. HLA-DRB1*15:01 is also correlated with more evident, diffuse spinal cord involvement and with higher disability score [10].
Distribution of alleles of MHC genes in different ethnicities varied in dependence on human population history primary due to geography, epidemics and other factors [11, 12]. In a Canadian pediatric cohort, the linked haplotype DRB1*15:01~DQA1*01:02~DQB1*06:02 was strongly associated with the Europeans and less strongly with non-Europeans [7]. Meta-analysis of 9 genotyping studies from China showed that HLA-DRB1*15 was associated with the risk of MS in the Chinese population, but significantly lower in Europeans [13].
Some factors other than HLA genes might direct the course of MS spectrum disorders. For example, in East London, MS risk was lower by 59% in Black and 85% in South Asian populations in comparison with English [14]. According to the study, Asian risk of MS is 80% lower than Caucasian. The incidence rate of MS in Japan increased from 1986 to 2013 from 8.1 to 18.6 cases/100,000 in period 1986-2013, but it is still significantly lower in comparison with European countries with 115 cases/100,000 population in 2015 [15]. The limited data from Kazakhstan showed MS prevalence as 10.1 per 100 000 population, 16.8 in Caucasians, and 4.9 in Asians [16].
Performing MHC class II gene study in Kazakhstan, where the population usually consists of Kazakhs and Russians, who are living in the same geographic environment, is very interesting from a scientific point of view but also has a practical clinical meaning. Currently, standard diagnosis approaches for MS diseases include brain and spinal MRI and additional investigations. Such investigations as oligoclonal bands and neurofilament light protein in CSF, also optical coherence tomography are hard to access in Kazakhstan. Moreover, patients and medical personnel have skepticism toward MS diagnosis. Therefore, the proof of having an immunogenetic predisposition of MCH class II gene status towards MS gives additional verification of the correct diagnosis.
Many environmental factors are important in neuroimmune diseases development [17-20]. A multicenter study in Canadian MS study showed an association among MS with the haplotype HLA-DRB1*1501, vitamin D insufficiency and previous infection with the Epstein-Barr virus (EBV) [20]. Vitamin D plays a protective role in the disease progression, as it can alter HLA DR autoantigen expression and presentation [21]. In Japan, MS was associated with obesity, smoking and specific dietary habits [22]. Recent data illustrated that immunopeptidomes coded by HLA-DR15 reproduce molecular mimicry with EBV triggering MS development [23]. HLA-”humanized” mice showed that the most prominent encephalitogenic target antigens implicated in human MS were determined by DQB1*06:02, rather than by the DRB1*15:01 [24-27].
Child neuroimmune diseases consist of postinfection, para-infection, inflammatory, demyelinated and sometimes brain necrotizing conditions with heterogeneous pathobiological mechanisms and clinical manifestations [28]. Differential diagnosis includes genetics (immunogenetics), biomarkers, high-resolution MRI, and ELISA or PCR for etiological (viruses) diagnosis [28-32]. Example of a relatively benign MS spectrum disease is acute disseminated encephalomyelitis (ADEM) [32, 33]. One of the rare, but extremely severe forms of the neuroimmune disease is Rasmussen Encephalitis. It is characterized by hemispheric autoimmune inflammation, triggered by viruses, with progressive seizures and brain hemiatrophy outcomes [34, 35]. There is no proved treatment for this disease, anti-TNF agents as Rituximab and intravenous immunoglobulin were used with some success [36]. However, the only proven treatment for termination of progressive, serial seizures is subtotal hemispherectomy [37].
Study of 31 Japanese patients showed an association of postinfection necrotizing encephalopathy with DRB1*09:01 and DQB1*03:03 [38]. Korean study of the disease determined encephalopathy affecting the bilateral thalami, pons, and midbrain in a symmetrical pattern; hemorrhage was in half of the patients and severe neurological deficits in 80% of infants [39].
One of the rarest and severe neuroimmune conditions is Devic’s neuromyelitis optica (NMO). The disease has a severe course with frequent relapses and rapid disability, usually unreversible neurological deficit, causing blindness in more than half of cases and often wheelchair assistance [40-43]. Prevalence and morbidity of NMO in Korean cohort were 2.56 and 0.73 per 100000 accordingly in 2010-2016 [43]. Previously NMO was considered as an MS variant [44, 45], but detection of specific anti-AQ4 antibodies and irreversible blindness proved NMO as separate disease [46-53]. The specific hallmark of the disease is MRI picture of longitudinally extensive transverse myelitis, which is never seen in MS [49, 50]. The anti-Aquaporine-4 (AQP4) antibodies are very specific for NMO, but in 25% of cases, anti-Myelin oligodendrocyte glycoprotein (MOG) antibodies [54, 55] are seen. Some biomarkers are also associated with NMO (glial fibrillary acidic protein and others) [56].
The treatment of NMO should be aggressive to stop progression. Usually steroids, plasma exchange, and intravenous immunoglobulin in high doses or subcutaneous immunoglobulin for long time are used. Immunodepressants are often used too. Nevertheless, there is no distinct effective therapy for NMO [40, 57].
In Kazakhstan, there is limited data about the prevalence of neuroimmune disorders in children. A single study showed that the general prevalence of pediatric MS was approximately 10.1 per 100 000 people. The male to female ratio was 1:2.5 [16]. DR-gene panel helps verify diagnosis of MS and related conditions, especially in pediatric patients and in cases when immune methods are not available in clinical practice (oligoclonal bands in CSF, etc.).
The aim of this study was to investigate whether genetic variants in the MHC region show an association with MS or other pediatric neuroimmune disorders in Kazakh population.
2. MATERIALS AND METHODS
All pediatric patients with a verified diagnosis of MS-spectrum disorder hospitalized at the Aksai Clinic, Kazakh National Medical University between 2015 and 2021 were included in this study. Clinical, neuroimaging and immunological investigations were performed for all of them. The study was conducted after the approval of the Ethical Committee of Kazakh National Medical University (n. 208, 29.04.2015 and n. 11, 23.10.2020). Parents of all patients provided the informed consent document.
Overall, 34 children (24 girls, 10 boys) with demyelination of the central nervous system aged 4 to 18 years were enrolled. 21 children were diagnosed with MS (11 Kazakh origin and 10 – Russian; 4 boys, 17 girls), 7 with leucoencephalitis (all Kazakh, 5 boys, 2 girls) and 6 with Devic’s neuromyelitis optica (all Kazakh, 1 boy, 5 girls).
The diagnosis of MS and other neuroimmune disorders was performed according to the latest version of McDonald's criteria [58]. Brain MRI was performed in all children with a 1.5 Tesla MRI scanner (Simens 1.5 Tesla Aero, 16 channels apparatus, Germany) in FLAIR regime, with gadolinium contrast. In patients with NMO, the amount of antibody to aquaporin 4 in the serum was tested by ELISA kits (Anti-AQP4 Kit, AEA582Hu, Cloud-Clone Corp., USA).
The genotyping of HLA loci was performed at the Atchabarov Institute for Basic and Applied Biomedical Research (Kazakhstan), after approval of the relevant Ethics Committee (n. 208, 29/04/2015). Data from the healthy Kazakh population (Kazakh Normal) were obtained from Kuranov et al. [59] and healthy Russian population (Russian Normal) from Boiko et al. [60]. The DNA was isolated according to a standard protocol [60]. The HLA-DRB1, HLA-DQA1, and HLA-DQB1 loci were detected by real-time PCR. The following specificities were tested: DRB1 * 01, 03, 04, 07, 08, 09, 10, 11, 12, 13, 14, 15, 16; DQA1 * 01:01, 01:02, 01:03, 02:01, 03:01, 04:01, 05:01, 06:01; DQB1 * 02, 03:01, 03:02, 03:04, 03:05, 04:01/04:02, 05:01, 05:02/05:04, 05:03, 06:01, 06:02-8. The number of alleles is equal to twice the number of patients.
2.1. Statistical Analysis
Qualitative data were presented as absolute numbers and percentages. Genetic data were compared between children with neuroimmune disorders and healthy populations using the Fisher test. A p-value < 0.01 was considered statistically significant. The relative risk (RR) was also calculated. Statistical analysis was performed by SPSS (version 16). The genetic data (group of alleles) of our patients and the population standard were calculated by direct counting and compared using the χ2 criterion in the Fisher test for small samples and relative risk was calculated (RR), p<0.01 was considered significant.
3. RESULTS
Overall, 34 children (24 females and 10 males) with neuroimmune disorders of central nervous system were enrolled. They were aged between 4 and 18 years old. Among them, 21 children were diagnosed with MS (11 Kazakh origin and 10 – Russian; 4 boys, 17 females and 4 males), 7 children with leucoencephalitis (all Kazakh origin; 2 females and 5 males) and 6 children with NMO (all Kazakh, 5 females and 1 male).
3.1. Clinical Characteristics of Children with MS
Out of the 21 children with MS, a total of 11 children were of Kazakh origin, while 10 children were Russians. The average age of debut was 14.3 ± 2.7 years old. They were affected by relapsing-remitting MS. There were 6 out of 21 patients (28.6%) who experienced 2 relapses, 8 out of 21 (38.1%) had 3 relapses, 4 out of 21 (19.05%) had 4 relapses and a single patient (4.8%) had 5 exacerbations. The mean number of relapses was 3.15 ± 1.09.
The average number of exacerbations during the follow-up period was 3.5±0.85. The Expanded Disability Status Scale (EDSS) mean value at the baseline was 1.62 ± 0.6. After 2-4 years, the mean value increased (2.05 ± 0.71, p = 0.001).
Symptoms at the debut were mainly optic neuritis (17 out of 21 children, 80.9%), limb ataxia (13 out of 21 children, 61.9%) and truncal ataxia (11 out of 21 children, 52.4%). After 2-4 years, the most common symptoms were limb ataxia (15 out of 21 children, 71.4%), pyramidal symptoms (14 out of 21 children, 66.7%) and truncal ataxia (12 out of 21 children, 57.1%). Mean number of relapses was 3.15±1.09; in 6 of 21 patients (28.6%) had 2 relapses, 8 of 21 (38.1%) children had 3 relapses, and 4 of 21 (19.05%) patients had 4 relapses and 1 (4.8%) had 5 exacerbations. Data are presented in Table 1.
| Clinical Symptoms | Debut | After 2-4 Years | p-Value |
|---|---|---|---|
| EDSS (Mean ± standard deviation, M±m) | 1.62 ± 0.6 | 2,05 ± 0.71 | 0.001 |
| Number of relapses (M±m) | - | 3.15 ± 1.09 | 0.001 |
| Optic neuritis | 17 out of 21 (80.9%) | 11 out of 21 (52.4%) | |
| Strabismus | 3 out of 21 (14.3%) | 1 out of 21 (4.8%) | |
| Limb ataxia | 13 out of 21 (61.9%) | 15 out of 21 (71.4%) | |
| Truncal ataxia | 11 out of 21 (52.4%) | 12 out of 21 (57.1%) | |
| Pyramidal symptoms | 10 out of 21 (47.6%) | 14 out of 21 (66.7%) | |
| Hemiparesis | 7 out of 21 (33.3%) | 9 out of 21 (42.9%) | |
| Paraparesis | 3 out of 21 (14.3%) | 4 out of 21 (19.1%) | |
| Hyperkinesis | 2 out of 21 (9.5%) | 1 out of 21 (4.8%) | |
| Mood disturbances | 6 out of 21 (28.6%) | 11 out of 21 (52.4%) | |
| Mild cognitive dysfunction | 0 | 2 out of 21 (9.5%) |
| MRI | T2-Active Lesions in the Brain, Number of Children (%) | Gadolinium Positive Lesions in the Brain, Number of Children (%) | T2-Active Lesions in the Spinal Cord, Number of Children (%) |
|---|---|---|---|
| Debut | 21 (100%) | 9 (42.9%) | 2 (9.5%) |
| After 2 – 4 years | 19 (90.5%) | 8 (38.1%) | 5 (23.8%) |
Brain MRI was performed on all 21 children. All of them showed T2-active periventricular and subcortical lesions after the first attack. Regarding gadolinium positive lesions, they were diagnosed in 9 out of 21 children (42.9%). After 2-4 years, T2-active lesions were diagnosed in the brain of 19 children (90.5%) while gadolinium positive lesions were seen in 8 children (38.1%). Spinal MRI showed T2-active lesions in 2 children (9.5%) at the debut and in 5 children (23.8%) after 2-4 years (Table 2).
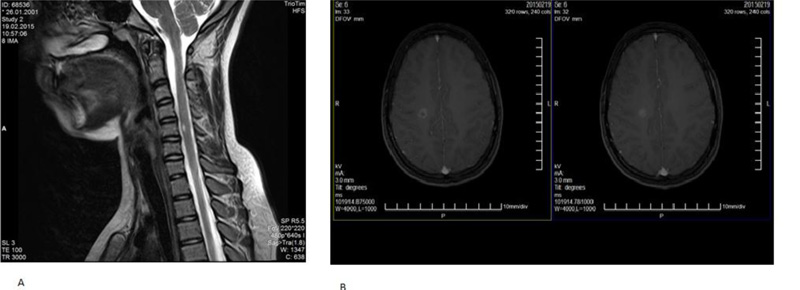
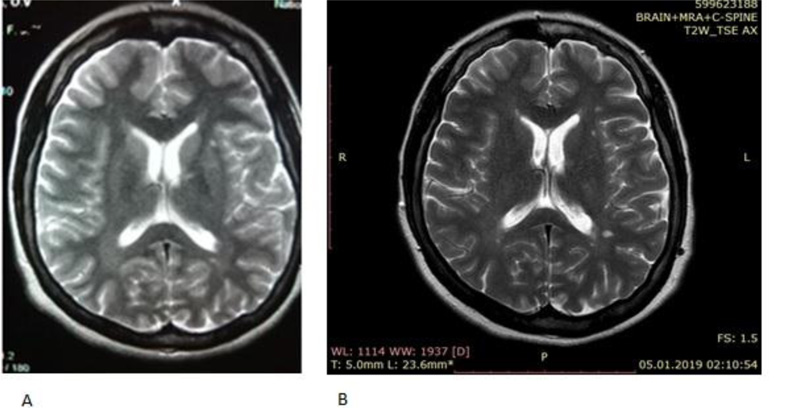
As an example, MS in one girl with severe MS (Picture 1) and with another girl with less progressive MS (Picture 2).
3.2. Clinical characteristics of children with leukoencephalitis
Of the seven children with leukoencephalitis, 4 suffered from Schilder’s type encephalitis, 2 from Rasmussen encephalitis and one suffered from multiphasic disseminated encephalomyelitis (Table 3). At the onset of the disease, the age was between 3 and 12 years (mean 8±4.2 years). Hyperthermia and drowsiness were diagnosed in all children with leukoencephalitis. Five children (71.4%) manifested headache and vomiting, while 3 children (42.9%) showed epileptic seizures (Table 3). One boy, who started the illness at 3 years old, had anamnesis of measles at 6 months (he died after 2,5 years because of the progression of the disease), so this likely was subacute sclerosing panencephalitis (SSPE); the other 4-years girl before the disease’s had the tick bite, and anti-togavirus IgG antibodies (she died after 3 years). In 2 children (boys) in anamnesis had perinatal persisted CMV infection, then after 5 years of age they presented with leukoencephalitis. The remaining 3 children during the debut of the disease were >10 years old and 2-4 weeks before the onset they had an upper respiratory viral infection.
| Clinical Symptoms | Debut, Number of Children (%) | After 2-4 Years, Number of Children (%) |
|---|---|---|
| Hyperthermia | 7 out of 7 (100%) | 2 out of 7 (28.6%) |
| Drowsiness | 7 out of 7 (100%) | 1 out of 7 (14.3%) |
| Headache | 5 out of 7 (71.4%) | 1 out of 7 (14.3%) |
| Epileptic seizures | 3 out of 7 (42.9%) | 2 out of 7 (28.6%) |
| Tetraparesis | 1 out of 7 (14.3%) | 1 out of 7 (14.3%) |
| Hemiparesis | 1 out of 7 (14.3%) | 1 out of 7 (14.3%) |
| Diffuse hypotonia | 2 out of 7 (28.6%) | 2 out of 7 (28.6%) |
| Hyperkinesis | 2 out of 7 (28.6%) | 2 out of 7 (28.6%) |
| Age of Debut | Gender | Aquaporin 4 Antibody (Normal < 1:10 Dilution of Serum) | |||
|---|---|---|---|---|---|
| At the Debut | After 1 Year | After 2-3 Years | |||
| 1 | 12 | Female | < 1:160 | < 1:80 | < 1:20 |
| 2 | 10 | Female | < 1:80 | < 1:40 | < 1:80 |
| 3 | 11 | Female | < 1:80 | < 1:40 | < 1:40 |
| 4 | 11 | Female | < 1:80 | < 1:40 | < 1:40 |
| 5 | 9 | Male | < 1:80 | < 1:80 | < 1:20 |
| 6 | 9 | Female | < 1:80 | < 1:40 | < 1:40 |
All children were treated with immunotherapy with intravenous IgG immunoglobulins and methylprednisolone every 2-3 months. Brain MRI showed T2-active lesions in all children, but not gadolinium positive lesions. After 4 years, one child developed symptomatic epilepsy, 3 children showed chronic fatigue, and 2 children died.
3.3. Clinical characteristics of children with neuromyelitis optica
In 6 children, the diagnosis of NMO was confirmed by the presence of serum AQP-4 autoantibodies, which were measured in the dilution of serum. Positive reference is more than 1:10 dilution, so here the maximum level was 1:160, the minimum - 1:20 (Table 4). Within the time, after one year, and after 2-3 years, all children, except one, showed reduced levels of autoantibodies, but anti-AQP-4 level did not achieve normal level (<1:10).
The clinical characteristics of children with NMO (Table 5) were distinguished by a significant vision loss, reflected in a distinct decrease of retinal nerve fiber layer (RNFL) thickness (p = 0.01). Moreover, there was an increase in neurological deficits, including paraparesis, seizures and urinary hesitancy.
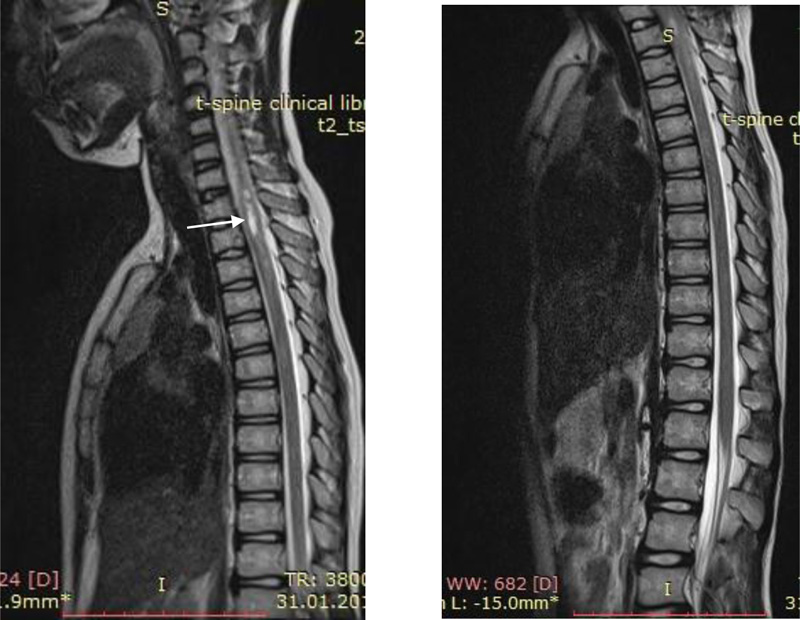
The spinal MRI performed in children with NMO showed longitudinal tranversus cervical and thoracic spinal demyelination. In Picture 3 it is shown dynamics of the spinal MRI of girl Zh. B. 2007 year of birth, with progressive loss of vision and spinal cord atrophy and severe paraparesis with urinary incontinence due to the NMO autoimmune necrotic process.
3.4. Genotyping of HLA loci
The results of genotyping for the genes of HLA loci in 21 children with MS (17 females and 4 males) are presented in Table 6. In Table 7, the children with MS are compared with a healthy population of Kazakhs and Russians.
| Clinical Characteristics | Debut | After 2-4 Years | p-Value |
|---|---|---|---|
| EDSS score (Mean ± standard deviation, M±m) | 2.7 ± 2.1 | 4.2 ± 2.1 | 0.01 |
| Loss of vision field (in % from normal volume of vision) | center, 30% | center, 67% | |
| Right, 34% | Right, 71% | ||
| Average RNFL total thickness (µm) | center, 84.4 ± 34.2 | center, 69.2 ± 26.2 | 0.01 |
| Right, 76.8 ± 40.9 | Right, 71.0 ± 38.0 | 0.01 | |
| Hemiparesis | 3 out of 6 | 3 out of 6 | |
| Paraparesis | 2 out of 6 | 3 out of 6 | |
| Epileptic seizures | - | 1 out of 6 | |
| Urinary hesitancy | - | 1 out of 6 | |
| Mood disturbances | 1 out of 6 | 3 out of 6 | |
| Cognitive dysfunction | 1 out of 6 | 2 out of 6 |
| Order Number of Patient | Age of Onset | Gender | DRB1 Allele | DQA1 Allele | DQB1 Allele | |||
|---|---|---|---|---|---|---|---|---|
| 1 | 12 | Male | 10 | 15 | 01:01 | 01:02 | 05:01 | 06:02-8 |
| 2 | 14 | Male | 09 | 14 | 03:01 | 01:01 | 03:03 | 05:03 |
| 3 | 16 | Female | 15 | 15 | 01:02 | 01:02 | 06:02-8 | 06:02-8 |
| 4 | 13 | Female | 15 | 15 | 01:02 | 01:02 | 06:02-8 | 06:02-8 |
| 5 | 17 | Female | 04 | 07 | 02:01 | 03:01 | 02 | 04:01/04:02 |
| 6 | 16 | Female | 13 | 15 | 01:02 | 01:03 | 06:02-8 | 06:02-8 |
| 7 | 16 | Female | 12 | 15 | 01:02 | 05:01 | 03:01 | 06:02-8 |
| 8 | 17 | Female | 03 | 15 | 05:01 | 01:02 | 02 | 06:02-8 |
| 9 | 14 | Female | 13 | 15 | 01:02 | 01:02 | 06:02-8 | 06:02-8 |
| 10 | 18 | Female | 03 | 11 | 05:01 | 05:01 | 02 | 03:01 |
| 11 | 14 | Female | 09 | 15 | 03:01 | 01:02 | 03:03 | 06:02-8 |
| 12 | 15 | Female | 04 | 15 | 03:01 | 01:02 | 03:02 | 06:02-8 |
| 13 | 17 | Female | 10 | 15 | 01:01 | 01:02 | 05:01 | 06:02-8 |
| 14 | 18 | Male | 03 | 11 | 05:01 | 05:01 | 02 | 03:01 |
| 15 | 11 | Female | 11 | 15 | 05:01 | 01:02 | 03:01 | 06:02-8 |
| 16 | 13 | Female | 07 | 15 | 02:01 | 01:02 | 02 | 06:02-8 |
| 17 | 12 | Female | 15 | 15 | 01:02 | 01:02 | 06:02-8 | 06:02-8 |
| 18 | 11 | Female | 09 | 15 | 03:01 | 01:02 | 03:03 | 06:02-8 |
| 19 | 14 | Male | 07 | 15 | 01:03 | 02:01 | 02 | 06:02-8 |
| 20 | 14 | Female | 09 | 15 | 03:01 | 01:02 | 03:03 | 06:02-8 |
| 21 | 15 | Female | 08 | 15 | 04:01 | 04:01 | 04:01/04:02 | 06:02-8 |
| Alleles | Kazakh Normal | Russian Normal | Children with MS | |||
|---|---|---|---|---|---|---|
| DRB1 | % | abs | % | abs | % | abs |
| *01 | 4.5 | 14 | 8.15 | 22 | 0 | 0 |
| *03 | 13.7 | 43 | 7.88 | 21 | 7.14 | 3 |
| *04 | 8.1 | 25 | 12.96 | 35 | 4.76 | 2 |
| *07 | 13.1 | 41 | 13.33 | 36 | 7.14 | 3 |
| *08 | 3.7 | 11 | 2.59 | 7 | 2.38 | 1 |
| *09 | 4.8 | 15 | 0.37 | 1 | 9.52 | 4 |
| *10 | 1.2 | 4 | 1.48 | 4 | 4.76 | 2 |
| *11 | 11.9 | 37 | 13.7 | 37 | 7.14 | 3 |
| *12 | 2.5 | 8 | 3.7 | 10 | 2.38 | 1 |
| *13 | 17.2 | 54 | 12.96 | 35 | 4.76 | 2 |
| *14 | 12.9 | 40 | 2.59 | 7 | 2.38 | 1 |
| *15 | 6.3 | 20 | 14.07 | 38 | 47.6 | 20 |
| *16 | 0.6 | 2 | 6.3 | 17 | 0 | |
| Number of alleles | 314 | 270 | 42 | |||
| RR of DRB1*15 | 13.36 | 5.55 | ||||
| p-value | < 0.001 | < 0.001 | ||||
| χ2 | 63.2 | 27.0 | ||||
As shown in Tables 6 and 7, the HLA-DR15 haplotype genes were detected in linkage disequilibrium (DRB1*15 ~DQA1*01:02~ DQB1*06:02) in 16 (76.2%) children with MS, or in 20 (47,6%) alleles. In comparison, the same pattern was seen in 6.3% of Kazakh Normal and 14.0% of Russian Normal. The relative risk (RR) for developing MS was more than 13 times higher than healthy Kazakhs (χ2 = 63.2; p < 0.001) and more than 5 times higher than healthy Russians (χ2 = 27.0, p < 0.001).
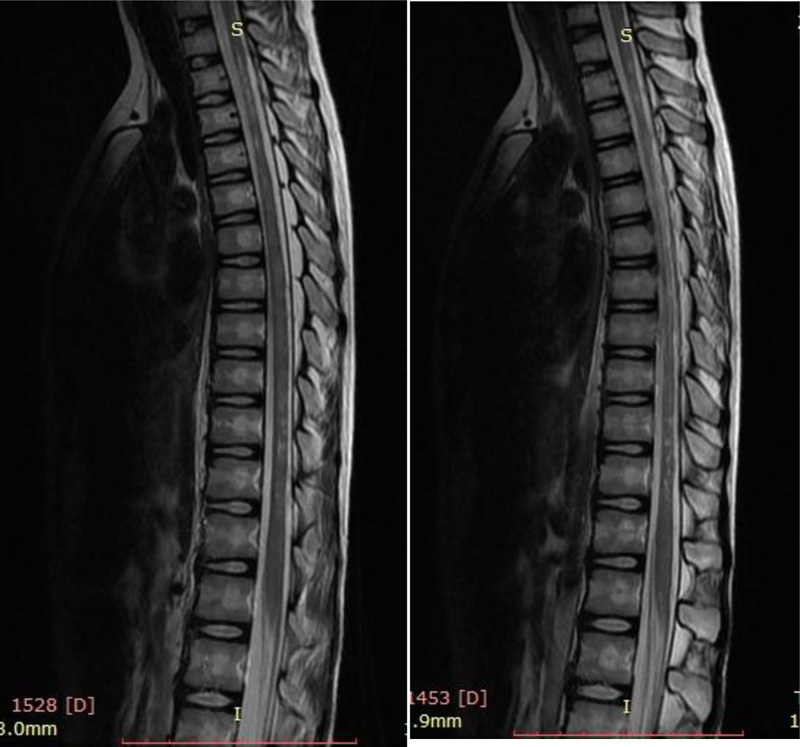
Children with DR15 (13 heterozygotes and 3 homozygotes) were predominantly females (15 out of 16 allele carriers). At the age of onset, they were younger (14.1 vs. 16.2 years old) Table 8. However, the difference was not statistically significant (p < 0.05). Three homozygous Russian girls manifested the disease at the age of 11, 12 and 16 years old. They showed a classical MS course with EDSS progression and distinct MRI features (Figs. 1-3).
The results of genotyping for the genes of HLA loci in 7 children with leukoencephalitis (2 females and 5 males) are presented in Table 9. The HLA-DR15 haplotype genes were detected in 3 out of 7 children and in 4 out of 14 alleles (28.6%) (RR = 5.88, χ2 = 9.74, p = 0.01 compared with Russian Normal). The allele DRB1*16 was found in two children (14.3%) (RR = 26, χ2 = 20.7, p = 0.01 compared with Kazakh Normal).
| Feature | HLA-DR15 allele | Other alleles |
|---|---|---|
| Number of mails | 1 | 3 |
| Number of females | 15 | 2 |
| Mean age (years) | 14.1±2 | 16.2±2.05 |
| p-value (age) | 0.045 | |
| p-value (gender) | 0.046 | |
| Order Number of Patient | Gender | DRB1 Allele | DQA1 Allele | DQB1 Allele | |||
|---|---|---|---|---|---|---|---|
| 1 | Male | 04 | 15 | 1:02 | 3:01 | 3:02 | 06:02-8 |
| 2 | Female | 15(02) | 16(02) | 01:02 | 01:02 | 06:02-8 | 05:02/4 |
| 3 | Female | 11(05) | 15(02) | 05:01 | 01:02 | 03:01 | 06:02-8 |
| 4 | Female | 09 | 11 | 03:01 | 5:01 | 3:02 | 3:03 |
| 5 | Male | 8 | 15 | 1:02 | 4:01 | 04:01/04:02 | 06:02-8 |
| 6 | Female | 03 | 10 | 0:501 | 01:01 | 02 | 05:01 |
| 7 | Female | 12 | 16 | 1:02 | 5:01 | 3:01 | 05:02/05:04 |
In children with NMO (5 females and 1 male), the HLA-DR15 haplotype genes were found in only one heterozygous child (16.7%) (RR = 2.94, χ2 = 1.02, p = 0.3 compared with Kazakh Normal) (Table 10).
| Order number of patient | Gender | DRB1 Allele | DQA1 Allele | DQB1 Allele | |||
|---|---|---|---|---|---|---|---|
| 1 | Female | 08 | 12 | 04:01 | 06:01 | 03:01 | 04:01/04:02 |
| 2 | Female | 01 | 13 | 01:01 | 01:02 | 05:01 | 06:02-8 |
| 3 | Female | 11 | 14 | 03:01 | 05:01 | 03:01 | 03:01 |
| 4 | Female | 14 | 14 | 01:01 | 01:01 | 05:03 | 05:02/4 |
| 5 | Female | 03 | 14 | 01:01 | 05:01 | 02 | 05:02/05:04 |
| 6 | Male | 09 | 15 | 01:02 | 03:01 | 03:03 | 06:02-8 |
The DRB1*14 allele was present in 66% of children with NMO (RR = 3.38, χ2 = 4.09, p = 0.05 compared with healthy Kazakhs).
4. DISCUSSION
In this study, we showed that children with MS group were characterized by a relapsing-remitting MS, with an EDSS progression from 1.62±0.6 to 2.05±0.71 in 2-4 years after debut. Primarily, about 90% of them were diagnosed with ADEM, as described in studies by other authors [12, 13]. The most prominent syndromes at debut were optic neuritis (80.9%) and cerebellar ataxia (61,9%), which were partly reversible.
Considering the HLA loci, there was a predominance of the haplotype DRB1*15 ~DQA1*01:02~ DQB1*06:02 as linkage complex. This pattern was detected in 20 (47.6%) of 42 DR-alleles, and in 16 (76.2%) patients from the investigation group. In comparison, in Kazakh Normal population, this haplotype was seen in 6.3% of investigated DR-alleles, in Russian normal – in 14.07%. In our cohort of children, the relative risk (RR) for developing MS was 13.4 times higher in Kazakh individuals and 5.6 times higher in Russian individuals. The haplotypes DRB1*15 ~DQA1*01:02~ DQB1*06:02 carriers were mostly female (15 from 16) and had a younger age of onset (14.1±2 against 16.2±2.05 years), similar to literature [5-7, 61-63]. In our previous studies, the haplotypes DRB1*15, DQA1*01:02 and DQB1*06:02 were detected in 32.2% of analyzed alleles [64]. This difference (47.6% vs. 32.2%) is related to the pool of data (number of enrolled patients). In the previous study, all children with a neuroimmune disorder were pooled together, MS and related conditions. In this study, we analyzed patients according to their exact clinical diagnoses.
Other numerous studies found that the haplotype DRB1*15~DQA1*01:02~ DQB1*06:02 was highly frequent in MS [2-5]. However, in African-Brazilian children with MS, the strongest association was observed with DQB1*06:02 rather than DRB1*15 [65]. Some authors suggested that these haplotypes did not predict the clinical course of MS [4]. Nevertheless, we described that the presence of DRB1*15 negatively influenced the follow-up of MS, as described by other authors, especially concerning worse brain MRI changes [6, 8, 66]. Varying MS diagnosis criteria, such as CSF studies and optical coherence tomography, are of limited access in Kazakhstan. Genetic studies of MCH class II genes help verify the diagnosis of MS spectrum diseases, which corresponds well to the findings of other MS study groups and help estimate the risks and evaluate the course of the diseases with increased accuracy.
In children with leukoencephalitis, the presence of haplotype DRB1*15~DQA1*01:02~ DQB1*06:02 was detected in 28.6% with RR=5,88. In Japan's study of post-viral acute necrotizing encephalopathy, the alleles DRB1*09:01 and DQB1*03:03 were approximately twice as frequent as controls [38]. Thereby, in our study, post-viral leucoencephalitis Rasmussen type in immune mechanism was closer to MS.
Children with NMO were characterized by vision loss and neurological deficits, including spastic hemiparesis and paraparesis. The genotyping analysis showed a high presence of DRB1*14 (seen in 66% of children with NMO with RR = 3.38), this allele has already been associated with systemic vasculitis in Japanese patients [68, 69]. In Muslim Arabs from Israel, individuals with anti-AQP4 NMO were characterized by a high presence of DQB1*04:02 and DRB1*10 [70]. Spanish NMO patients had DRB1*010 allele association compared with MS; further, these patients had DRB1*03 allele association in comparison with healthy controls [71]. According to data, genetically NMO is closer to systemic immune diseases rather than MS.
CONCLUSION
Thus, this study showed the appearance of haplotype DRB1*15~DQA1*01:02~ DQB1*06:02 in Kazakhstan patients as one of the relevant diagnosis criteria for MS and related conditions which is described in numerous MS DR-gene studies [4-8]. This helps Kazakh neurology specialists and parents of patients to have an accurate diagnosis and admit target immune disease modified treatment (beta-interferon, Capaxon and others) early.
Currently, in our country, such kind of treatment is available through mandatory medical insurance from the age 18. Usually, the start of treatment is delayed by 6-9 months due to bureaucratic procedures. Our study shows the need to initiate such treatment as soon as possible after establishing the diagnosis in children and adolescents because it is known that axonal losses and reduction of brain tissue volume in patients with child MS onset occur early and in bigger volumes till a certain age (for example by 40 years) [3, 5].
Early diagnosis and timely target treatment prevent further brain volume loss and irreversible EDSS alterations. Further study would admit more patients and incorporate deep neurological and immunological studies of MS and related conditions.
LIST OF ABBREVIATIONS
| CNS | = Central Nervous System |
| NMO | = Neuromyelitis Optica |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was conducted after approval of the Ethical Committee of Kazakh National Medical University (n. 208, 29.04.2015 and n. 11, 23.10.2020).
HUMAN AND ANIMAL RIGHTS
No animals were used for studies that are the basis of this research. All the humans used were in accordance with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Parents of all patients provided the informed consent document.
STANDARDS OF REPORTING
STROBE guidelines were followed.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
This research was financially supported by the grant AP09562785 (Creation and implementation of a modern system of diagnostics, monitoring and treatment of neuroimmune orphan diseases in children in the Republic of Kazakhstan, registration number 0121РК00600, provided by the Ministry of Education, Republic of Kazakhstan).
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
ACKNOWLEDGEMENTS
We express our sincere gratitude to Dr. A. B. Kuranov and the Research Institute of Fundamental and Applied Medicine named after B. Atchabarov for the help and support received in conducting the study. We would like to thank TopEdit (www.topeditsci.com) for its linguistic assistance during the preparation of this manuscript.


