All published articles of this journal are available on ScienceDirect.
Anxiety-related Endophenotypes and Hazardous Alcohol Use in Young Adults are Associated with a Functional Polymorphism in the SLC6A4 Gene
Abstract
Background:
A functional polymorphism (5-HTTLPR, rs4795541) in the serotonin transporter (SLC6A4) gene has been shown as an important candidate for several psychiatric and behavioral traits.
Objective:
The objective of this study was to examine the possible interaction of this polymorphism with physical neglect in childhood on the presentation of anxiety traits and hazardous alcohol consumption in young Colombian subjects.
Methods:
272 young adults (mean age: 21.3 years) were evaluated with the Childhood Trauma Questionnaire, the Zung Self-rating Anxiety Scale, the Big Five Inventory, the Cohen’s Perceived Stress Scale, the Alcohol, Smoking, Substance Involvement Screening Test and the Alcohol Use Disorders Identification Test. Genotyping for the 5-HTTLPR polymorphism was carried out using conventional PCR. A linear regression model, corrected by age and gender, was used.
Results:
We found that individuals with the L/L genotype showed higher scores on physical neglect (p=0.0047), anxiety symptoms (p=0.028), neuroticism (p=0.019) and perceived stress (p=0.035). L/L genotype was a risk factor for hazardous alcohol use in young adults (OR=3.06, p=0.0003). No GxE interactions were observed in our data.
Conclusion:
Our results provide novel evidence for the role of a functional polymorphism in the SLC6A4 gene on the relationship of childhood trauma, anxiety-related traits and risky consumption of alcohol.
1. INTRODUCTION
Anxiety Disorders (AD), which include panic disorder, generalized anxiety disorder, post-traumatic stress disorder, phobias, and separation anxiety disorder, are among the most common mental disorders around the world [1, 2]. These disorders usually have an early onset followed by a chronic course, being the sixth leading cause of disability worldwide [3]. The current global prevalence of anxiety disorders is 7.3%, suggesting that one in 14 people has an anxiety disorder at any given time [1]. The prevalence is higher in women than in men, with women to men ratio of 1.9:1 and it has been remained relatively constant across time [4]. Prevalence estimates for AD were significantly higher in people exposed to an armed conflict [1] and, in this context, the Colombian population has suffered from one of the longest internal armed conflicts in the world [5]. According to the DSM-V, substance use disorders are characterized by at least two of the diagnostic criteria (such as social and legal problems related to use, among others) [6] and are important contributors to the global burden of disease [7]. Hazardous alcohol use has been defined as a pattern of consumption that leads to a higher risk of harmful effects for the individual [8].
AD and hazardous alcohol use are complex traits with a complex etiology. They both have an environmental and a genetic component. It is expected that the combination of several genetic polymorphisms interacts with environmental variables to increase the risk [9-11]. Even though several genes are associated with both disorders, the SLC6A4 gene, which encodes the serotonin transporter, became a major candidate gene for AD and hazardous alcohol use [10, 12]. It is interesting in the context that several drugs of psychiatric use, such as paroxetine and citalopram, are inhibitors of this transporter [12].
The most commonly studied polymorphism in this gene is the non-coding variant 5-HTTLPR (rs4795541), which is located in the promoter region [13]. This polymorphism is a Variable Number Tandem Repeat (VNTR), composed of 24 bp repeats which generate different alleles. The two main alleles are the short (S) with 14 repeats and the long (L) with 16 repeats [13].
Functional studies have reported that the L allele is associated with higher levels of transcriptional activity and with a higher rate of serotonin uptake [14]. In addition, several works have shown that the S allele, in general, is associated with a less favorable response to pharmacological treatment in affective and anxiety disorders [15, 16].
Lesch et al conducted the first study that found an association between the SLC6A4 promoter polymorphism and anxiety-related traits, including the neurotic personality trait [17], and the authors reported that the 5-HTTLPR poly-morphism accounted for 3-4% of population variability in neuroticism [17]. In other studies, it has been reported that individuals with the S/S genotype scored higher for neuroticism on the big five personality model. In addition, in patients with depression and anxiety disorders it has been observed a higher frequency of the S allele [18, 19]. The 5-HTTLPR polymorphism has been investigated in the context of stressful life events, including childhood maltreatment, showing association with the increase in the susceptibility to depressive and anxious symptoms [20]. Subjects with both the S/S genotype and with high levels of childhood maltreatment have been shown to have a higher sensitivity to anxiety [20].
There is a growing evidence for the association of genes involved in the serotonergic system with hazardous alcohol use [10]. A recent meta-analysis showed that the S allele is associated with an increased risk for alcohol dependence [21]. In addition, other studies have found that subjects with the S allele reported more frequent drinking [22-24]. In contrast, other works have found an association with hazardous drinking in subjects with the L allele [25].
The purpose of this study was to examine the hypothesis that the 5-HTTLPR polymorphism might interact with physical negligence in childhood on the risk for the presence of anxiety symptoms and hazardous alcohol consumption in young adulthood.
2. METHODS
2.1. Participants
Two hundred seventy-two Colombian young adults were included in this study (21.3 years of age, SD=3.8, 75% women) [26, 27]. Participants were enrolled in medical and nursing schools and were invited to participate in the study [28, 29]. All participants signed a written informed consent and this study was approved by the Institutional Ethics Committee of the Antonio Nariño University. The procedures performed were in accordance with the ethical standards of the insti-tutional research committee and with the 1964 Helsinki declaration and its later amendments.
A self-report instrument including general demographic and medical information was applied and it served to identify subjects with personal history of neuropsychiatric disorders. Ages over 18 years and absence of personal history of neuropsychiatric disorders were the inclusion criteria for the study (12 subjects were excluded due to it). All the phenotypic data were collected through self-report instruments, which were applied by a psychologist. The included individuals were mainly from low and medium socioeconomic status (34 and 45%, respectively), had mainly secondary as their education level (79%) and single was the most frequent marital status (93%).
2.2. Psychological Measurements
To evaluate physical negligence, the Childhood Trauma Questionnaire (CTQ)-brief version was used [30]. This questionnaire is a 28-item self-administered inventory that provides an assessment of child abuse and neglect through five dimensions: physical, sexual and emotional abuse and physical and emotional neglect. Scores of 9 (for both men and women) are indicative of physical abuse and physical neglect and of 5 and 7 for sexual abuse (in men and women, respectively) [31]. The CTQ has adequate psychometric properties in different languages, included the Spanish [31]. In the current study, the Cronbach´s alpha was 0.89.
Anxiety-related endophenotypes were measured using the Big Five Inventory (BFI-S) [32], in the short version (15 items), the Zung Self-Rating Anxiety Scale (ZSAS) [33] and the Cohen’s Perceived Stress Scale (PSS) [34].
The BFI-S inventory measures 5 dimensions of personality: Factor O (Openness), Factor C (Conscien-tiousness), Factor E (Extraversion), Factor A (Agreeableness) and Factor N (Neuroticism) on a 7-point Likert scale: from 1 (strongly disagree) to 7 (strongly agree) [32]. In our analysis, we only included the factor N (Neuroticism) because it is one of the most established risk factors for anxiety and mood disorders [35]. The Cronbach’s alpha was 0.62 for Neuroticism in the current study.
ZSAS is a self-report questionnaire designed to measure anxiety symptomatology in the general population. It is composed of 20 items with 4 possible responses: (1) never, (2) rarely/sometimes, (3) frequently and (4) always [33]. Each item is scored from 1 to 4 according to the severity of anxiety symptoms and total scores range between 20 to 80 points. ZSAS has shown to have a good internal consistency in Colombia [36]. In the current study the Cronbach’s alpha was 0.85.
The PSS-10 items [34] is a self-report scale used to assess an individual’s experience of general perceived stress over the past month. The scores of this scale range from 0 to 40, higher scores mean higher levels of stress. It has been validated in Colombia population [37]. In the current study, the Cronbach’s alpha was 0.85.
Hazardous alcohol use was measured with two different scales, searching for a better characterization of the con-sumption:
The Alcohol, Smoking and Substance Involvement Screening Test (ASSIST) [38] is an instrument created by the World Health Organization (WHO) for the detection of risky or abusive consumption of substances. This instrument assesses the frequency, patterns of substance use and risky use of tobacco, alcohol, amphetamine-type substances, cannabis, cocaine, hallucinogens, inhalants, opioids, sedatives and other substances through eight questions. Total scores for each substance are categorized as ‘low-risk use’ (0-3), ‘moderate-risk use’ (4-26) or ‘high-risk use’ (>26) [38]; it has been validated in different languages, included Spanish [39]. In this study, alcohol was the substance with higher consumption (42.3%) and for that reason we selected hazardous alcohol use as one of the outcome variables and their Cronbach’s alpha was of 0.75.
The Alcohol use disorders identification test (AUDIT) [8] is a brief written screening method to identify current harmful and hazardous drinking in primary care settings, in both developing and developed countries. The 10-item scale was designed to assess three conceptual domains: drinking, behavior/dependence and alcohol-related problems. It has been validated in a Colombian sample [40]. The cut-off point used in the current study was ≥8 for risk of hazardous alcohol use, based on the recommendations by the WHO [8]. In the current sample, the Cronbach´s alpha was 0.78.
2.3. Genotyping
The extraction of DNA was performed by a salting out method, using 400 µl of peripheral blood [41]. For the genotyping of the 5-HTTLPR polymorphism (rs4795541), a conventional PCR was carried out in a Labnet MultiGene 96-well thermal cycler (Labnet International Inc, Edison, NJ, USA). The PCR reaction was performed in a total volume of 20 µl, with 20 ng of DNA, 0.8 U of Taq polymerase (Bioline, London, United Kingdom), 10 µM of each primer (F: 5´ATG CCA GCA CCT AAC CCC TAA TGT 3´, R: 5´GGA CCG CAA GGT GGG CGG GA 3´), 0.25 mM of each dNTP, 10X reaction buffer and 1.5 mM MgCl2. The PCR program consisted in one cycle of 94 oC for 30 s, 35 cycles of 94oC for 30 s, 66 oC for 30 s, 72oC for 40 s, and a final step of 1 cycle of 72 oC for 5 min. The PCR products were allowed to run for 40 min at 140 V, in a 2% agarose gel. The genotypes were determined as follows: L/L: 419/419 pb, L/S: 419/375 bp, S/S: 375/375 bp. With the aim of verifying the genetic results, 10% of the samples were randomly selected and re-analyzed.
All mandatory health and safety procedures were accomp-lished in all the experiments carried out in this study.
2.4. Statistical Analysis
The psychometric analysis of the scales and questionnaires was performed using standard approaches: Internal consistency through Cronbach’s alpha and factorial structure of the tests using exploratory factorial analysis.
Normal distributions from the psychological scales scores were examined through skewness and kurtosis and bivariate correlations with Pearson Coefficient were used. These statistical analyses were performed with the Statistical Package for the Social Sciences (SPSS v. 18)
A linear regression model corrected by age and gender, using SNPStats program, was used to identify the possible association of the SNP with anxiety symptoms, perceived stress, physical neglect, hazardous alcohol use and neuroticism [42]. SNPStats program was also used to calculate genotype and allelic frequencies and Hardy-Weinberg equilibrium. The best genetic model was selected based on the Akaike Information Criterion [42]. For the current study, the Bonferroni corrected p value (0.05 divided by 6), was 0.008.
In addition, multiple regression models were carried out to examine GxE Genotype (G) and Environment (E) interactions:
(A) Physical neglect in childhood x 5-HTTLPR genotypes to predict anxiety symptoms
(B) Physical neglect x 5-HTTLPR genotypes to predict neurotic trait
(C) Physical neglect x 5-HTTLPR genotypes to predict perceived stress
(D) Physical neglect x 5-HTTLPR genotypes to predict hazardous alcohol use, for both AUDIT and ASSIST alcohol subscale.
To know whether 5-HTTLPR genotype played a role as mediator, the mediation-in-serial models were employed, using multiple regressions, following the procedures recommended by Hayes [43]. The mediation effect occurs if the association between the Independent Variable (IV) and the Outcome Variable (DV) is eliminated (complete mediation) or reduced (partial mediation) when the Mediator Variable (MV) is introduced in the analysis. The following mediation models were carried out:
- Physical neglect in childhood was inserted as an IV, anxiety-related phenotypes were inserted as DVs separately and 5-HTTLPR genotype was inserted as the MV.
- Physical neglect was inserted as an IV, alcohol use was inserted as DV for AUDIT and ASSIST alcohol subscale, separately and 5-HTTLPR genotype was inserted as the MV.
Direct, indirect and total effects, standard errors and 95% confidence intervals (biased-corrected bootstrapped; 1000= repetitions) for each model, were calculated using speci-fication model 4 of the PROCESS plug in (V.2014) [43] in SPSS (v. 18).
3. RESULTS
In our sample, the factorial structures found were the expected: six-factor solution for CTQ (64.1% of variance), five-factor solution for BFI-S and ZSAS (62.1% and 56% of variance respectively), two-factor solution for PSS (56% of variance), one-factor solution (46.3% of variance) for alcohol use (ASSIST) and three-factor solution for AUDIT (59% of variance).
The allele frequencies in our sample were 0.51 and 0.49 for the S and L alleles, respectively. The S/S genotype was observed in 26% (n = 70), the S/L genotype in 50% (n = 136) and the L/L genotype in 24% (n = 65) of the subjects in the current sample. The genotype frequencies were in Hardy-Weinberg equilibrium (p = 1).
Correlations were significant only between physical neglect and anxiety symptoms (r = 0.278, p<0.01) and physical neglect and perceived stress (r = 0.221, p<0.01), no association was found between physical neglect and neurotic trait (r = 0.113, p>0.05), or for physical neglect and ASSIST alcohol subscale (r = 0.100, p>0.05) or AUDIT scores (r = 0.103, p>0.05).
We found significant associations between behavioral variables and 5-HTTLPR: anxiety symptoms (p = 0.029), physical neglect (p = 0.0047), alcohol use (p = 0.011), perceived stress (p = 0.035) and neuroticism (p = 0.019) (Table 1). The results for association with physical neglect were significant after a Bonferroni correction for multiple testing.
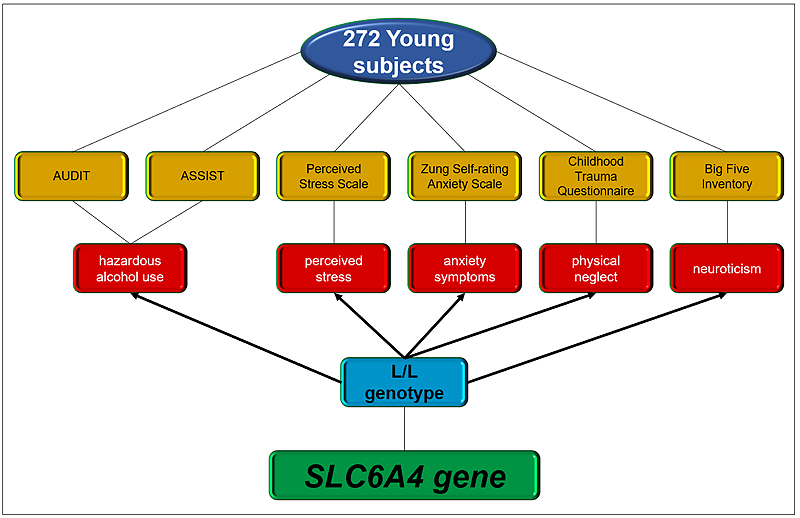
The risk of alcohol use based on recommended cut-off points in AUDIT (81 subjects for moderate risk) and ASSIST (150 subjects for moderate risk) was associated significantly with 5-HTTLPR polymorphism. The odds ratio was higher for carriers of the long variant of the 5-HTTLPR polymorphism (AUDIT, OR= 2.01, CI: 1.04 – 3.87; ASSIST alcohol subscale, OR= 3.06, 1.62 – 5.79) (Table 2). Results for the ASSIST alcohol subscale were significant after a Bonferroni correction for multiple testing. An overview of the associations found in our work is shown in Fig. (1).
No GxE interactions were observed in the data, for the multiple regression models, as well as for neither of the models of mediation analysis (Table 3). 5-HTTLPR genotype was excluded of all the regression models for anxiety symptoms, neurotic trait and perceived stress (as outcomes variables) and 5-HTTLPR genotypes and physical neglect in childhood (as predictors) (p = 0.467, p = 0.088, p = 0.809 respectively). For hazardous alcohol use models, physical negligence was excluded from both models (p = 0.397 for AUDIT and p = 0.871 for ASSIST alcohol subscale).
| Measurements | – | Genotypes | Mean Scores (SE) | p value |
|---|---|---|---|---|
| Anxiety symptoms | – | S/S –S/L | 37.8 (0.6) | 0.028 |
| – | L/L | 40.5 (1.3) | ||
| Physical negligence | – | S/S – S/L | 6.3 (0.1) | 0.0047* |
| – | L/L | 7.2 (0.3) | – | |
| Hazardous alcohol use | ASSIST scores | S/S – S/L | 6.6 (0.5) | 0.011 |
| – | L/L | 9.2 (0.9) | – | |
| AUDIT scores | S/S – S/L | 4.6 (0.3) | 0.25 | |
| – | L/L | 5.31 (0.6) | – | |
| Neuroticism | – | S/S – S/L | 4.1 (0.1) | 0.019 |
| – | L/L | 4.47 (0.1) | – | |
| Perceived stress | S/S – L/L | 18.81 (0.6) | 0.035 | |
| – | S/L | 17.32 (0.5) | – |
| Scale | Genotype | Risk use | OR (95% CI) | p value | |
|---|---|---|---|---|---|
| Low | Moderate | ||||
| AUDIT | L/S – L/L | 134 (70.4%) | 67 (82.7%) | 2.01 (1.04 – 3.87) | 0.03 |
| S/S | 56 (29.5%) | 14 (17.3%) | 1.0 | – | |
| ASSIST | S/S – L/S | 104 (86%) | 102 (68%) | 1.0 | – |
| L/L | 17 (14.1%) | 48 (32%) | 3.06 (1.62 – 5.79) | 0.00033* | |
| Multiple Regression | Mediation Analysis | ||||||||
| – | Physical neglect in childhood | 5-HTTLPR interaction | 5-HTTLPR interactiona | ||||||
| – | B | 95% CI | p | B | 95% CI | p | Effect | 95% CI | p |
| Neurotic trait | 0.155 | 0.01-0.11 | 0.001 | 0.103 | -0.02-0.37 | 0.088 | 0.002 | -0.002-0.012 | >0.05 |
| Anxiety symptoms | 0.281 | 0.52-1.25 | 0.001 | 0.043 | -0.97-2.11 | 0.467 | 0.007 | 0.009-0.084 | >0.05 |
| Perceived stress | 0.23 | 0.26-0.77 | 0.001 | 0.014 | -0.94-1.20 | 0.809 | 0.001 | -0.010-0.043 | >0.05 |
| Hazardous alcohol use (AUDIT) | 0.05 | -0.01-0.02 | 0.397 | 0.093 | -0.16-1.32 | 0.128 | 0.007 | -0.006-0.040 | >0.05 |
| Hazardous alcohol use (ASSIST) | 0.01 | -0.01-0.02 | 0.871 | 0.176 | 0.04-0.20 | 0.004 | 0.006 | -0.016-0.079 | >0.05 |
In the mediation models, 5-HTTLPR genotype was not significant mediator for any relationship between variables (Table 3).
4. DISCUSSION
The findings of the present study show an association between physical neglect in childhood, anxiety-related phenotypes, hazardous alcohol use and 5-HTTLPR poly-morphism (Fig. 1). Our results suggest that 5-HTTLPR polymorphism could be an important risk factor for hazardous alcohol consumption and anxiety-related endophenotypes, such as the neurotic trait, anxiety symptoms and perceived stress in young adults. However, we did not find GXE interactions between 5-HTTLPR polymorphism and physical neglect to predict anxiety-related phenotypes or hazardous alcohol use.
Childhood maltreatment is an important risk factor for affective disorders and a major public health problem [44]. Exposure to emotional and physical neglect is associated with increased risk for a wide range of behavioral problems, including anxiety disorders, alcohol abuse, depression and suicidal behavior [45].
Previous findings have shown that many potential mechanisms may explain the mentioned association between neglect in childhood and increased risk for mental health problems and alcohol consumption. One of the most plausible explanations is that the neurobiological development can be altered by the maltreatment during childhood, which in turn can negatively affect a child's cognitive, emotional, and social growth, leading to behavioral and learning problems that can persist throughout life [44]. Moreover, it is widely accepted that in many cases some victims of childhood maltreatment may try to manage the subsequent distress through the use of alcohol, tobacco, medicaments and other drugs [46]. In addition, epigenetic studies have proposed that early life events affect DNA methylation of the SLC6A4 gene [47, 48].
Previous findings have shown that the risk for affective disorders is elevated among carriers of the S allele when they were exposed to stressful life events [49]. However, our data suggest that the L/L genotype is likely to be important on psychosocial development, particularly in those settings that increase the risk of anxiety symptoms, neurotic trait and hazardous alcohol use. These findings are consistent with some studies that revealed an interaction between the L allele and the increased risk of depression in subjects exposed to stress [25]; however, few studies have replicated these findings in healthy young adults with measures of anxiety symptoms [25].
To date, there are few studies that have investigated the association of the functional 5-HTTLPR polymorphism with hazardous alcohol use in healthy subjects. While some studies showed evidence of an association between 5-HTTLPR and psychological variables related to hazardous alcohol use [50], others found a significant result with higher alcohol intake [22-25]. Our findings of a dominant model for the association of the S allele and hazardous alcohol use is supported by previous articles [23, 51]. In addition, our finding of heterosis in the association between HTTLPR and perceived stress is concordant with a previous study [52].
Multiple structural neuroimaging studies have shown that young adults with history of heavy drinking have lower volumes in prefrontal cortex [53], with some of these changes being partially reversible [54]. In addition, several functional neuroimaging studies have shown that these young adults also have elevated levels of brain activity in fronto-parietal areas during cognitive tasks and increased levels of neural responses in several mesocorticolimbic areas, such as the anterior cingulate cortex, the amygdala and the striatum [53]. Animal models have also found that alcohol exposure affects neurochemical processes in mesocorticolimbic brain regions [55].
In accordance with the above findings, the association of the 5-HTTLPR polymorphism with childhood maltreatment and anxiety-related phenotypes is not clearly understood and the reports of association have been inconsistent and difficult to replicate [56]. Our results have further shown a protective role for the S allele in the risk for alcohol consumption and the development of anxiety symptoms and neuroticism. Our findings are supported in part by previous results where S/S genotype of the 5-HTTLPR polymorphism appeared to be protective against depressive symptomatology in a supportive early context [57]. However, several studies have reported the opposite effect, with the S/S genotype being a risk factor for affective disorders [49].
A possible contributory factor of this inconsistency may be heterogeneity in the measurements and characteristics of environmental adversity investigated in the different studies, particularly, the nature of the stressors studied. For example, the duration of exposure to adversity and the type of stressors studied [58]. Recall bias in the psychological assessment might be another cause of inconsistent findings; it is widely accepted that retrospective reports on traits or experiences are often discrepant (in comparison with prospective studies). In many cases, individuals may attempt to mentally reconstruct information about past events, personality trait or common behaviors, by generating detailed reports based on their general beliefs [59]. In patients with anxiety disorders, such as panic disorder and obsessive-compulsive disorder, it has been documented a negative recall bias in retrospective self-reports, resulting in a retrospective overestimation of the disorder-specific symptoms [60].
CONCLUSION
In the current study, two types of stress were found as associated with the SLC6A4 gene: chronic stress and acute stress, through physical neglect in childhood and perceived stress over the past month respectively; but only results for physical neglect were significant after a Bonferroni correction for multiple testing. This is consistent with the results of a meta-analysis [18] that found that the effect of 5-HTTLPR polymorphism (S allele) on neurotic trait is only apparent when those carrying the short allele are exposed to the stressful environment and with the results of another meta-analysis [58] that reported robust evidence for 5-HTTLPR moderating the relationship between both childhood maltreatment and specific stressors and depressive symptoms.
Current understanding of the specific characteristics of the stressors that are moderated by 5-HTTLPR is insufficient [58]. However, considering previous data and our current results, specific and chronic stressors (such as childhood maltreatment) might be more important than acute stressors in the relationship of 5-HTTLPR polymorphism and anxiety related-traits.
Limitations of the current study include the analysis of a single polymorphism in the SLC6A4 gene and the use of a convenience sample design, which includes a higher proportion of females.
To our knowledge, the current study is the first work that explores the relationship between 5-HTTLPR polymorphism and anxiety-related phenotypes and hazardous alcohol consumption using validated scales for several psychosocial variables (Table S1) [18, 58]. Furthermore, this is the first study of this type in a Latin American sample.
FUTURE PERSPECTIVE
In futures studies, it could be important to examine the relationship between 5-HTTLPR polymorphism and exposure to environmental adversity, using different phenotypic characterizations as well as different types of adversity. In this context, future longitudinal studies, involving functional neuroimaging methods, will provide an interesting approach for analyzing the cumulative effect of genetic factors, early alcohol use and childhood trauma on brain function.
AUTHORS' CONTRIBUTIONS
KMJ participated in acquisition and analysis of genetic data and drafting and critical revision of the manuscript. AJP participated in study design, acquisition and analysis of psychological data and drafting and critical revision of the manuscript. SL-L participated in analysis of genetic data and drafting and critical revision of the manuscript. AA participated in analysis of psychological data and drafting and critical revision of the manuscript. D.A.F participated in study design, acquisition and analysis of genetic and psychological data, drafting and critical revision of the manuscript.
ETHICS APPROVAL AND CONSENT TO PARTI-CIPATE
The study was approved by the Institutional Ethics Committee of the Antonio Nariño University.
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experi-mentation (institutional and national), and with the 1964 Helsinki declaration and its later amendments.
CONSENT FOR PUBLICATION
Written informed consent has been obtained from all the participants.
AVAILABILITY OF DATA AND MATERIALS
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
FUNDING
This study was supported by a research grant from Colciencias (grant # 823-2015) and by a grant from the Spanish Ministry of Economy, Industry and Competitiveness PSI2015-65026 (MINECO/FEDER/UE).
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors thank to Andres Camargo, who assisted with recruitment and evaluations of subjects.
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publishers Website along with the published article.


