All published articles of this journal are available on ScienceDirect.
Facial Palsy: A Retrospective Study of 416 Cases Based on Electrodiagnostic Consultation
Abstract
Background:
Facial nerve palsy (FP) is a frequent neurological condition caused mostly by Bell´s Palsy (BP).
Objectives:
The main objective of this study is to describe electrophysiological parameters in a retrospective 28-year review of 416 cases of FP based on electrodiagnostic consultation.
Methods:
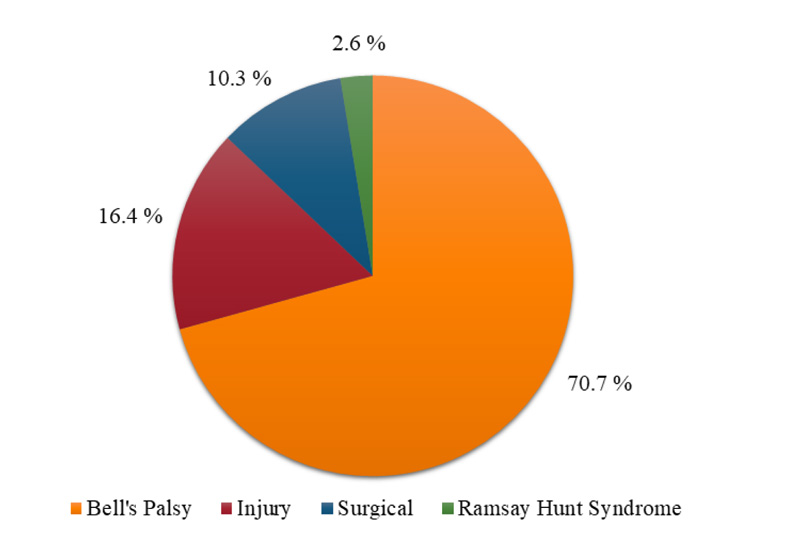
In total, 520 exams from 416 patients over a 28-year period were reviewed. Sex, age, etiology, comorbidities, and variables from electroneurography and needle electromyography were analyzed. Cases were grouped as BP (70.7%), injury (16.4%), iatrogenic (10.3%) and Ramsay Hunt syndrome (RHS) (2.6%).
Results:
The mean age was 41 years (3-82), 53.4% female. Diabetes was the most frequent comorbidity. Estimated Axon Loss (EAL), >90%, was found in 50% of the cases, mainly in the iatrogenic group. The amplitude drop of the Compound Muscle Action Potentials (CMAPs) was proportional in the Orbicularis Oculi, Orbicularis Oris and Nasalis muscles. The absence of CMAPs was more frequent in the iatrogenic group and less frequent in the BP one. Bell´s palsy associated with diabetes was more severe. The R1 latency (blink reflex) was significantly longer in the BP group (P>0.001). Synkinesis due to the misdirection of regenerating axons was much more frequent in the BP and RHS groups.
Conclusion:
Bell´s palsy was the most common cause. The EAL was equal in all facial branches. Facial nerve inexcitability was more frequent in the iatrogenic/injury groups. The R1 latency was found to be prolonged in the BP group and the only good prognosis indicator in a few cases. Misdirection reinnervation was more frequent in BP and RHS groups. There was no sex or side predominance.
1. INTRODUCTION
Acute peripheral facial nerve Palsy (FP) is most frequently idiopathic (Bell's Palsy or BP) and represents 46.4% to 69.2% of all FPs [1-4]. The described annual incidence of BP is 20 to 30 [5], and 32 per 100,000 people [2]. Other causes are represented by (i) iatrogenic, most after ear, parotid or vestibular schwannoma surgery in 14.4 to 17.4%, (ii) trauma in 8.9 to 10.5%, (iv) herpes zoster infection (Ramsay-Hunt syndrome or RHS) in 7.5%, and (iv) miscellaneous in 5.1% [3, 4].
The facial nerve main branches are 1. greater superficial petrosal nerve, which leaves the main trunk at the site of the geniculate ganglion; 2. parasympathetic nerve supply for the lacrimal gland (by way of an anastomosis between the lacrimal nerve and the zygomatic nerve), for the nasal mucosa, and for the palate; 3. stapedius nerve (disproportionally increased loudness or hyperacusis and by a painful sensation or phonophobia produced by acoustic stimuli of higher intensities); 4. chorda tympani: fibers responsible for taste sensation at the so-called anterior two-thirds of the tongue and, preganglionic secretory fibers originating from the upper salivatory nucleus and synapsing in the submaxillary ganglion innervating the salivary glands; and 5. digastric branch, which leaves the main trunk below but close to the stylomastoid foramen, immediately below the posterior auricular nerve which supplies the muscles of the auricle [6, 7].
Extracranial lesions are the most common FP topography and cause weakness in all the nerve branches to the muscles. Sensory loss in the external auditory canal, auditory fluttering due to weakness of the stapedius muscle, ageusia, and involvement of the salivary and lacrimal glands associated with facial weakness, suggest intracranial injury [2]. This differential diagnosis is relatively easy, and most patients receive correct diagnoses in emergency services [8].
Steroids are typically used in BP, RHS, and traumatic facial palsy. All these facial palsy etiologies are associated with inflammation and swelling of the facial nerve [9]. Specifically for BP, steroid administration is the most effective therapy and should be prescribed early, preferentially within three days of the onset [9-11]. According to Nam et al. [9], low-dose steroid therapy (1 mg/kg/day oral prednisolone) is highly recommended in patients with acute facial palsy. The combination of antiviral medication with steroids appears to have only a modest benefit for BP [10-13]. For FP without recovery up to 12 months from the onset, surgery with nerve transfers (neurotization) can be done. Trigeminal motor neurotization, involving the Masseter muscle branch without graft interposition, is an effective procedure with minimal residual chewing difficulty. The restoration of the mouth rhyme and smile is the main goal [14].
Facial electroneurography (ENoG) or nerve conduction study were introduced by Esslen [15] (1973) and is still the best-established method to estimate the prognosis in the first days from the onset. It offers a major advantage over other electrical tests of providing an objective, quantitative assessment of facial nerve degeneration. Using the healthy side as a reference, the reduction in amplitude of the Compound Motor Action Potential (CMAP) on the affected side is thought to represent the percentage of nerve fibers which have degenerated [16].
Electrical stimulation of the facial nerve at the mandibular angle and recording at the Orbicularis Oculi (OOc), Nasalis, Orbicularis Oris (OOr) and Frontalis muscles show the CMAP amplitude values to be compared to the opposite side [17]. A severe axonal loss has a poor prognosis, even for long-term evaluations [18]. Later, needle Electromyography (EMG) can reveal small amplitude and short duration Motor Unit Action Potentials (MUAPs), typical for ongoing reinnervation [18, 19]. Finally, the Blink Reflex (BR) evaluation of the R1 latency, by stimulating the supraorbital nerve and recording from the OOc muscle, allows the entire efferent peripheral pathway of the facial nerve to be studied on the affected side [19, 20]; the R1 potential is absent in the presence of a transmission block [21]. It is also very useful to rule out diffuse demyelination (e.g., autoimmune neuropathies).
The aim of this study is to retrospectively analyze the electrodiagnostic evaluation (EDX) data from all FP patients over a period of 28 years. The electrophysiological parameters studied were the CMAP amplitude (absolute and comparative values) for prognostic evaluation, R1 latency from BR, and MUAPs recorded from facial muscles through needle electrode for the evaluation of reinnervation.
2. MATERIALS AND METHODS
We retrospectively analyzed a cohort of patients with FP extracted from our EDX database at the Hospital de Base, Sao Jose do Rio Preto, Sao Paulo, between February 1989 to April 2017. The variables studied were age, sex, referred specialty, etiology, comorbidities, and date of onset. Four groups were formed: 1. Bell's palsy; 2. FP after nerve injury; 3. Iatrogenic FP; and 4. Ramsay Hunt syndrome. Most of the EDX were performed by JAK or by another board-certified doctor. The machines used over time were an IBM-4E (USA), a Dantec (Denmark) and a Portable Keypoint (Medtronic, Denmark).
The electrophysiological parameters obtained at the seventh day from the onset were; the CMAP latency (ms) and amplitude (mV) obtained after the facial nerve stimulation at the mandibular angle while recording in the OOc, OOr, and Nasalis muscles on both sides, and the latency of R1 of the BR by stimulation of the supraorbital nerve (trigeminal) and recording in the OOc (facial nerve) muscle on both sides.
The EMG was evaluated in the OOc, OOr and Frontalis muscles and included the following: subjective quantification of fibrillations and positive sharp wave potentials, morphology (normal, ongoing reinnervation, or chronic reinnervation) and recruitment pattern (normal or reduced) of the MUAPs, and the presence of synkinesis as a result of misdirection of the facial motor axons into distinct regions during reinnervation. The EMG is particularly useful after the third week from the onset to evaluate ongoing reinnervation.
The inclusion criteria consisted of patients who had EDX in our institution and presented the word “facial” in the report conclusion. Exclusion criteria: incomplete or conflicting ENoG/EMG results (37 cases), hemifacial spasm (9), nontemporal association with self-reported otitis (8), systemic disorders without a precise correlation (6), congenital abnormality not well-defined (5), isolated distal facial branch abnormality (4), Guillain-Barre syndrome (4) and a localized aesthetics procedure complication (1). Also, 104 patients who had more than one EDX evaluation were counted as one for the epidemiological analysis. In a few reports, when only the conclusion was shown, the epidemiological variables were included.
We used descriptive statistics that included mean and standard deviations for variables with a normal distribution (Gaussian) and medians and percentiles for variables with non-normal distributions. Comparisons of groups, when possible, were conducted by 1. chi-square tests for categorized variables and 2. t-tests for continuous variables. Minitab software was used.
3. RESULTS
The database search for “facial” in the EMG/ENoG report conclusion counted 594 patients. After exclusion, 520 exams from 416 patients were included. Facial palsy etiological frequencies are shown in Fig. (1).
Demographic variables (age and sex) related to the four etiological groups are shown in Table 1. In the injury group, 50% of the cases were caused by traffic accidents involving motor vehicles (cars, motorcycles, trucks), bicycles, or pedestrians versus motor vehicles collision. In the iatrogenic group, neoplasms represented 44.2% of those cases.
The seasonal frequency of 215 BP cases that had the precise date of onset was as follows: autumn (March to May), 30.7%, winter, 18.5%, spring, 27.4%, and summer, 23.3%. The comparison of the proportions showed more cases in autumn than in winter (p-value = 0.0034) and summer (p-value = 0.0284). Neurology (51.4%) and otorhinolaryngology (27.7%) were the most frequent referring medical specialties. Right FP was found in 47.6% of the cases, left in 50%, and bilateral in 2.4%. The mean and median values between the onset of symptoms and the EDX evaluation were 348 and 60 days, respectively (range: 5 to 8,760 days). This information was not available for 98 patients.
Diabetes Mellitus (DM) was the most commonly associated disorder and found in 6.5% of cases. In BP cases, DM was found in 25 cases (8.5%) with a mean age of 56 years (range: 29 to 81 years). In 60% of those cases, CMAPs were absent, all of which were assessed before 240 days from the onset. Among the RHS cases, DM was found in 9.1%. In the iatrogenic group, DM was found in 2.3%. Among the 297 patients with unilateral FP from the BP and RHS groups, 17 (5.7%) had the previous FP. In this group of 17 patients, there was a good recovery in 64.7%, there was no information for 29.4%, and there was residual motor dysfunction in 5.9%. More than one exam was performed in 104 patients, but it was not possible to perform a statistical analysis regarding the outcome due to the non-standardization and the small sample.
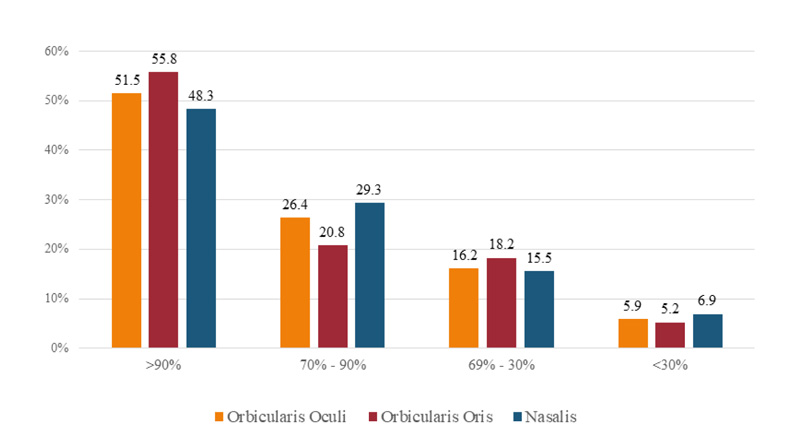
The facial nerve CMAP amplitude values with recording in the OOr, OOc, and Nasalis muscles from 243 patients who had ENoG performed within 7 to 180 days of FP onset are shown in Fig. (2).
| Group | n | % | Age (mean/SD) | Range | Female |
|---|---|---|---|---|---|
| Bell’s Palsy | 294 | 70.7 | 42 ± 17.6 | 3 - 82 | 57.1% |
| Injury | 68 | 16.4 | 33 ± 17.6 | 3 - 70 | 27.9% |
| Iatrogenic | 43 | 10.3 | 46 ± 17.4 | 5 – 79 | 67.4% |
| Ramsay Hunt syndrome | 11 | 2.6 | 45 ± 17.6 | 15 - 76 | 63.6% |

Comparative analysis of the CMAP amplitude values (affected versus nonaffected side) for the Estimated Axonal Loss (EAL) is shown in Fig. (3). There was no EAL significant difference among all regions analyzed ENoG (OOc, OOr, and Nasalis muscles). In all cases, the EAL was proportional in temporal, zygomatic and mandibular/buccal facial branches without a statistically significant difference even if we graded it as severe (90 to 100% loss), moderate (70 to 90% loss) or mild (<70% loss). Additionally, in the four etiological groups, there was no evidence of disproportionate EAL.
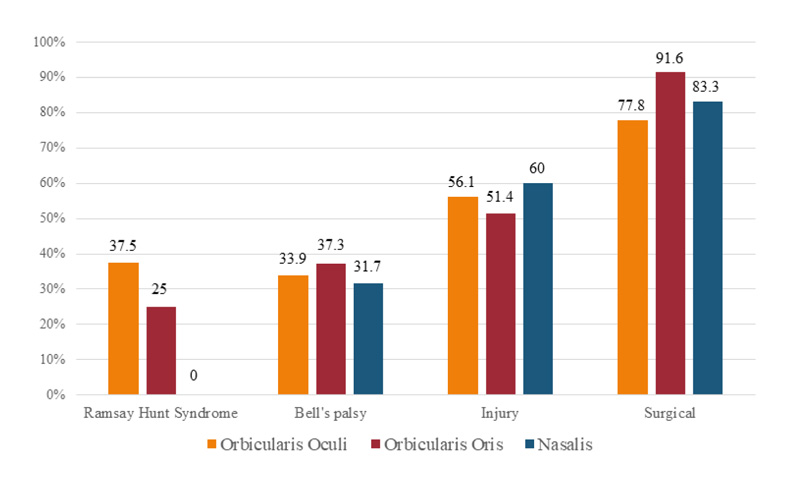
The complete axonal loss, meaning no CMAP was obtained in OOr, OOc and Nasalis muscles in the first 180 days, was found in 20.1% of RHS, 34.3% of BP cases, 55.8% of injury cases and 84.2% of iatrogenic cases Fig. (4). There were statistically significant differences in BP versus injury (p=0.0114), BP versus iatrogenic (p<0.0001), RHS versus iatrogenic (p=0.0029), and injury versus iatrogenic (p=0.0026).
Fig. (5) shows the four FP etiological groups and the percentage of cases in which CMAPs were inexcitable (complete axon loss) from recording in the OOc, OOr and Nasalis muscles in the first 180 days after onset in patients who presented misdirection reinnervation (synkinesis) after 180 days. Post-facial paralysis synkinesis was not found or it was less common in the injury and iatrogenic groups and very common in the BP and RHS groups. These frequencies do not represent the outcome of the same cases, but simply the referral for electrophysiological studies.

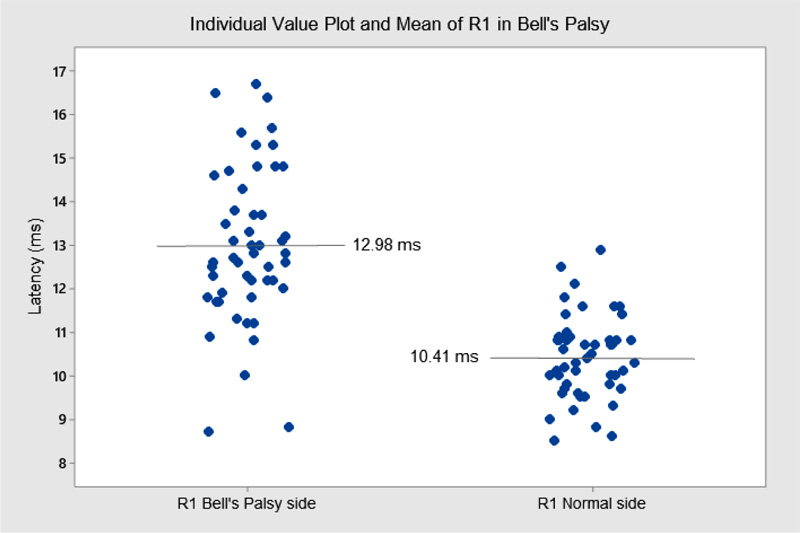
The R1 latency (BR) significantly increased in 91 BP cases Fig. (6). The presence of DM increased, even more, the R1 latency (13.8 ms versus 13.0 ms). An interesting finding was the absence of CMAP in the OOc muscle and R1 from BR present in 4/27 cases (14.8%). The R1 latency analyzed in 10 cases from the iatrogenic and injury groups showed a 12.4 ms mean latency versus 10.1 ms on the nonaffected side (P = 0.2528). Another interesting finding was the CMAP absence in the OOc muscle in 4 of those cases; despite the absence of a CMAP, we recorded some MUAPs by EMG.
4. DISCUSSION
Similar to the current published literature, this study found that BP was the most frequent FP cause (70.7%), followed by injury (16.4%), iatrogenic (10.3%) and RHS (2.6%). In other studies, BP represented to be 46.4% to 69.2% [1-4], iatrogenic 14.4 to 17.4%, trauma 8.9 to 10.5%, and RHS in 7.5% [3, 4]. Bell's palsy accounted for almost 90% of all nontraumatic or iatrogenic FP cases [22]. The high frequency of injury here probably was related to motorcycle accidents before Brazil's law requiring all motorcycle drivers and riders to wear a helmet [23, 24].
Bell's palsy occurred similarly in both sexes (57.1% female and 42.9% male) with a mean age of 42 years (range: 3 to 82 years), and affected equally both sides of the face, as described by others [1, 8, 25-26-27]; more cases occurred in the autumn, in spite of the literature pointed out no definite season predominance [2, 27]. Bilateral BP occurred in 2.4% of the cases, without reference to an associated disease. In other reports, a bilateral presentation was found in 0.3 to 2.0% [28]. Cases who reported more than one BP episode (recurrence) represented 5.7%. The incidence of recurrent facial palsy has been reported to range from 2.6 to 15.2% [2, 8, 25, 26]. In our cases, there was a favorable recovery of the first episode in 64.7% similar to 72.2% described by Chung et al. [29]. There is no relation to prognostic deterioration due to BP recurrence, but a positive familial history increases this possibility by 2.5 times [30]. The comorbidity most commonly found in our BP cases was DM, representing 8.5% of the cases, less than the 11.2% found by Yanagihara & Hyodo [31]. In this subset, the mean age was higher and the electrophysiological parameters indicating a severe prognosis were significantly more frequent, 60% versus 32.7%, as already described by others [2, 32, 33]. Even diabetic patients having an incomplete facial paresis, the recovery was very poor, with only 25% achieving normal facial muscle function [2]. Due to the frequent association with peripheral neuropathy, R1 latency is more prolonged than in BP cases without DM [34]. The clinical severity of facial palsy does not depend on DM duration [34].
Ramsay Hunt syndrome is due to infection or reactivation of the Herpes Zoster virus in the geniculate ganglion, and in our cases, the frequency was below that described in the literature (2.6% versus 15%) [35]. The ratio BP to RHS was 15:1 [2], so the expected RHS would be 20 cases instead of the 11 described here. Peitersen [2] stressed that only two-thirds of the patients had blisters localized in the ear, proving that it is necessary to inspect the head, neck, oral cavity, pharynx and thorax, and if the vesicles do not occur before or simultaneously to the FP, probably it will be considered as BP. Ramsay Hunt syndrome was more frequent in females (63.6%) and with an average age of 45 years. Despite the small sample, DM was more frequent in the RHS group (9.1%), but according to the literature, this high percentage is not true. In patients over 40 years of age, the rate of association of DM and RHS was only 1.3% [31].
The injury group was younger (mean age of 33 years), mostly male (72.1%), and the FP was frequently secondary to motor vehicle accidents. Guerrissi [36] reported good recovery when early signs of reinnervation occurred in cases with an extratemporal injury. Patel & Groppo [37] reported 15 to 20% of FP in cases associated with a temporal bone longitudinal fracture.
The iatrogenic group was older (mean age of 46 years) and mostly women (67.4%). Intracranial causes were for resection of neoplasms and other space-occupying lesions secondary to chronic infection related to ear/mastoid. Extracranial causes were mainly for parotid tumor resection as identified by Dib et al. [38]. In cases of chronic otitis media associated with temporal and/or mastoid bone destruction, the facial nerve may be involved [39], but in most cases, a cholesteatoma was presented [40] requiring surgical intervention [41]. We could not associate otitis and FP in this study since the temporal relationship between the two events was not precise in our cases.
FP with a bilateral presentation is usually associated with systemic diseases [22, 42-43-44] namely, post-influenza, infectious mononucleosis, HIV infection, Hansen's disease, Lyme disease (also Bannwarth lymphocytic meningoradiculitis, which is associated with ticks in Europe), Guillain-Barre syndrome variants with facial diplegia and paresthesia [45], syphilis, HTLV-1 infection, DM, acute porphyria, acute leukemia, sarcoidosis, amyloidosis, and multiple sclerosis [28]. Bell's palsy could also present as facial diplegia [28].
In our FP cases, the CMAP amplitude obtained from OOc, Nasalis and OOr muscles, representing distinct facial nerve branches, had about the same percentage values for absent CMAP, <0.50 mV CMAP and, >0.51 mV CMAP in the first 6 months from onset. The EAL by the CMAP amplitude comparison, health versus weak side, is well-defined and the best prognostic criterion at least for the first 23 days, when the maximal asymmetry value is reached [21]. In our cases, the EAL >90%, 70-90%, 30-69%, and <30% groups were similar for the OOc, Nasalis and OOr muscles. The absolute (mV) and relative (mV difference in percentage) from CMAP amplitude values were equivalent for all three muscles. Engström et al. [46] had already demonstrated it for Frontalis, OOc, Nasalis and perioral - Mentalis, Depressor Labii Inferioris, OOr and Depressor Anguli Oris muscles. Our findings confirm that the damage to facial motor axons was always proportional for all branches, meaning that the facial nerve fascicles are poorly separated and all of them respond equally to aggression.
The EAL 100% (CMAP absent) in the first 180 days from the onset was found in 77.8 to 83.3% in the iatrogenic group, the most severe one, and in 31.7 to 37.3% in the BP group, the mildest one. In spite of those findings, cases with EAL 100% leading misdirection reinnervation (synkinesis) after 180 days were found in all RHS cases, 68.7% of BP cases, 12.5% of iatrogenic cases and, none from injury cases. Cases due to compressive, inflammatory or infectious disorders with severe initial EAL tend to recover more frequently with facial synkinesis probably due to the poorly fascicular distribution of axon bundles into the nerve trunk, allowing misdirection reinnervation. In contrast, cases due to injury aggravation, either direct or secondary to surgery, mostly caused severe damage or disruption without the possibility of any reinnervation, either normal or misdirection [4]. Our findings were not the same from others, where 56% of synkinesis were found in the traumatic group and 33% of the cases were Bell’s palsy [47].
It is unquestionable, the EAL prognostication value for FP in the early days from the onset. However, many issues are disputed among different series. 1. What is the optimal day for the first exam? Complete axonal degeneration (Wallerian degeneration) takes about 9-10 days to occurs distally to the injury. However, for FP, the segment is shorter compared to other trunk nerves from limbs. Chow et al. [48], chose to establish ENoG values 5 to 14 days after the onset of facial palsy as a prognostic indicator. Medeiros et al. [49], found the maximum amplitude reduction for estimated axonal loss in the sixth day from onset. Baugh et al. [50], reported that with complete paralysis, electrophysiologic testing results become stable, and therefore informative, approximately 7 days following symptom onset. Azuma et al. [51], emphasized that the value of ENoG 10 to 14 days after the onset of facial palsy is a predictive factor for the development of facial synkinesis. Lee [18] concluded that ENoG is useful to predict the prognosis only during the first 2 weeks after the onset. For Ardiç et al. [52], the importance of ENoG begins on the third day after and lasts until the eighth day in 80% of the patients. We understand that there is no reason to use the ENoG at the very 3 days after the onset, and we suggest using it from the 7th day on; in this time, there is no chance to further reduction in CMAP amplitude. 2. What is the normal variability from right/left side for CMAP comparison? It is strongly dependent on the examiner’s experience to fix the electrode in the correct place equally on both sides. The average normal variation shown by the intra-individual CMAPs between both sides of the face was 25% [53] and between 26% to 39% [54]. So, differences over 30% could be found in healthy subjects. 3. What is the test-retest variability for correct interpretation of many exams in the same patient? This variability was described as 3.15% [53], 6.2% [17], and 10% [55, 56]. So, even if a 10% difference in the follow-up exams is found, we cannot be 100% secure about improving or worsening. 4. Finally, what is the magic percentage value for prognostication using the EAL? Most of the authors use 90% EAL [50] as the limit for the good (less than 90%) and bad prognosis (90% or more), since it was described by Esslen [15] and also by Fisch [55]. Smith et al. [16], using 90% limit, found a complete recovery in all but one with EAL <90%. Kitisomprayoonkul et al. [47], found 75% of BP patients with BP <90 EAL gained complete recovery, and all cases of BP with <70% EAL had a complete recovery. According to a multivariate analysis, a patient with >90% EAL has a 7.5 times greater probability of a non-satisfactory recovery in the long term [57]. Patients with an ENoG with 53.5% EAL have a high risk of developing synkinesis [51]. According to logistic regression analysis, the best predictive indicator of poor recovery is the EAL >50% assessed in the frontalis muscle [20]. ENoG with EAL >90% has the poorest prognosis [5]. The complete recovery rate was found to be significantly higher when the EAL <80% the OOc and <65% for the OOr [58]. ENoG with an EAL ≥ 90% was found to be more frequent in subjects who developed synkinesis before palsy recovery, whereas subjects with an EAL from 60 to 90% showed a high risk of late synkinesis onset [59]. The critical ENoG EAL value to identify nonrecovery patients after 1 year was calculated to be >81.5% in BP and 78% in RHS cases [60]. Bell’s palsy or RHS, who have more than 85% EAL on ENoG, may have a poor prognosis [33]. For absolute values, when the CMAP amplitude was >0.5 mV, complete recovery with no residual deficits is expected in 2-6 weeks [61].
Between 1 to 3 months from the onset, EMG will be more helpful. There is an increased risk of sequelae if regeneration has not begun in this interval [52]. The detection of spontaneous fibrillation is a reliable sign predicting unfavorable outcome [1]. Some have more confidence in EMG, especially when performed serially [4].
The BR reflex (R1 latency values) is less frequently used as a prognostic tool in FP [19] because it is more frequently absent than CMAPs. However, in 4 patients of our series, CMAPs were not obtained in the OOc muscle, and an R1 with increased latency was found in the same muscle. In those cases, there was a good recovery. A comparison of the R1 latency values in BP showed a significant increase in the affected side compared to the nonaffected side (13.0 versus 10.4 ms). The presence of the R1 action potential alone, regardless of latency value, is a good prognostic variable. Very high R1 latency values are useful in identifying potential autoimmune or hereditary demyelinating neuropathies. Finally, the BR is also useful in chronic cases with post-facial paralysis synkinesis (misdirection reinnervation) since the stimulation of the supraorbital nerve evokes the R1 response not only in the OOc muscle but also in the OOr muscle [62]. In the injury and iatrogenic groups, the R1 component was present in 38.5%, there was no statistically significant increase in latency compared to the opposite side, and in 4 of these 10 cases, the CMAP was absent with MUAPs in the OOc muscle, thus showing an early sign of recovery.
CONCLUSION
We have concluded that the FP etiology is comprised of BP (70.7%), injury (16.4%), iatrogenic (10.3%) and RHS (2.6%). The CMAP amplitude proportionally decreased in OOc, OOr and Nasalis muscles, regardless of the estimated axonal loss. The percentage of severe cases was much higher in iatrogenic cases, followed by injury, RHS and BP. 4. The R1 latency (for the BR) was statistically prolonged only in BP cases and the presence of R1 represented, in a few cases, the only electrodiagnostic parameter of a better prognosis. Moreover, misdirection reinnervation with facial synkinesis was much more frequent in BP and RHS cases. Last, there was no side or sex predominance.
LIMITATIONS OF THE STUDY
This study mostly did not follow the same patients from the FP onset to a specific endpoint time. This case series was analyzed retrospectively based only on our EDX data from the last 28 years. So, the patients did not follow a protocol for EDX timing after FP or a clinical protocol for each EDX visit. Thus, some bias was inevitable: some epidemiological, etiological and electrophysiological data were missed. It was not possible to evaluate the detailed neurological examination or the treatment history of these patients. For all cases, the EDX operator was a highly specialized medical doctor, board-certified. Even with these limitations, the etiological groups were reliably formed and the electrophysiologic parameters were highly trustworthy.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Research Ethics Committee of the FAMERP.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The dataset generated and analyzed during the study is with the corresponding author can be made available on a reasonable request.
FUNDING
This study was supported by a grant (2017-14665-5) from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP): medical student (VFMF) and mentor (JAK).
CONFLICTS OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGMENTS
Declared none.


